Magnesium
 |
 |
 |
|
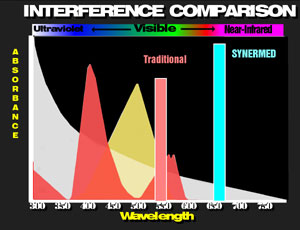
The Synermed® magnesium is detected in the infrared region of the spectrum thus avoiding the absorbance of hemoglobin and minimizing the absorbance of lipemia. Except in geriatric populations, most pathologic magnesium values are below normal. Consequently, signal levels [absorbance changes] are very low. Low levels of serum background noise is very important to avoid chromatic interference, especially in the important pathologic low levels.
-
-
-
-
- Single Liquid-Stable Ready-to-Use Reagent
- Minimal Interference from Serum Bilirubin, Hemoglobin and Lipemia
- Extended "On-Board" Stability
-
-
-
Principles of the Reaction
Magnesium reacts with Xylidyl Blue I to produce a Magnesium-xylidyl blue complex which is red in color at the alkaline pH of the reagent. The disappearance of the near-infrared absorbing blue color is proportional to the amount of magnesium present in the serum.
Xylidyl Blue I + Mg+2 -> Xylidyl Blue-Mg+2 complex
(blue) (red)
The disappearance of the initial blue color as the xylidyl-Mg complex is formed can be measured between 630 nm and 670 nm and is proportional to the magnesium concentration. Calcium is prevented from forming a complex with the xylidyl blue by the inclusion of EGTA in the reaction mixture.
Analytical Range
Most applications of this procedure have been found to be linear to 2.5 mmol/L (5.0 mEq/L) magnesium.
Special Performance Characteristics
- The Synermed magnesium methodology was correlated to a widely accepted colorimetric method. The calculated linear regression on 37 samples ranging from approximately .65 to 2.14 mmol/L with Synermed on the Y-axis was Y = 0.95X + 0.05 with a correlation coefficient of 0.950.
- The sensitivity of the procedure is such that an absorbance change of 0.001 will detect as little as 0.011 mmol/L magnesium.
- The within run precision of the method as applied to an automated analyzer was determined by assaying 30 samples of quality control materials at two levels of magnesium concentration yielding the following results:
Mean SD CV 0.7 mmol/L 0.010 1.5% 2.3 mmol/L 0.013 0.6% - The run to run reproducibility of the method as applied to an automated analyzer was determined from the values obtained by 5 replicate analyses of quality control material assayed over a 22 batch period.
Mean SD CV 0.69 mmol/L 0.01 1.4% 2.25 mmol/L 0.05 2.2%
Expected Results
Decreased levels of serum magnesium are seen in a variety of pathologic conditions. The most common reason for lowered levels in otherwise normal patients is alcoholism. Causes of hypomagnesemia are classified below.
-
- Gastrointestinal and nutritional causes
- Prolonged parental fluid administration without magnesium
- Prolonged severe diarrhea, e.g. ulcerative colitis, regional enteritis, and chronic laxative abuse
- Intestinal malabsorption
-
-
- Idiopathic steatorrhea
- Tropical sprue
- Short-bowel syndrome from any cause
-
-
- Alcoholism
- Acute and recurrent pancreatitis
- Starvation with attendant metabolic acidosis
- Diabetic ketoacidosis
- Protein-calorie malnutrition including kwashiorkor
- Renal causes
- Prolonged use of diuretics
- Renal disease
-
- Renal tubular acidosis
- Recovery from acute tubular necrosis
- Chronic glomerulonephritis and pyelonephritis
- Familial magnesium wastage
- Gentamycin-induced renal injury
-
- Endocrine and metabolic causes
- Hyperthyroidism
- Hyperparathyroidism with osteitis fibrosa cystica
- Malacic bone disease with hypercalcemia
- Primary and secondary aldosteronism
- Excessive lactation
- Congenital hypoparathyroidism
- Infant born of mother with hyperparathyroidism
- Neonatal and childhood causes
- Infantile convulsions
- Newborns of diabetic mothers
- Genetic (male) hypomagnesemia
- Exchange transfusions
- Gastrointestinal and nutritional causes
Hypermagnesemia and intoxication occur mainly in patients with serious renal insufficiency and in eclampsia when large doses of magnesium salts are administered. The administration of magnesium-containing antacids in patients with renal failure is an important cause of magnesium intoxication.
Connect with us
Certifications
 |
 |
 |
 |
||

